FDA-cleared for Foley, straight, and Malecot indications — engineered to remove the source of infection and restore patient comfort.
The Foley catheter hasn’t fundamentally changed since 1933. The balloon-based retention mechanism, the structural cause of trauma, infection, and patient discomfort, has remained unchallenged. Until now.
Lotus® is the first and only urinary catheter FDA 510(k)-cleared for the same indications as Foley, Malecot, and straight catheters in a single device — designed to improve patient outcomes and reduce total cost of care.
The future of catheters starts
now.
evolve

| Metric |
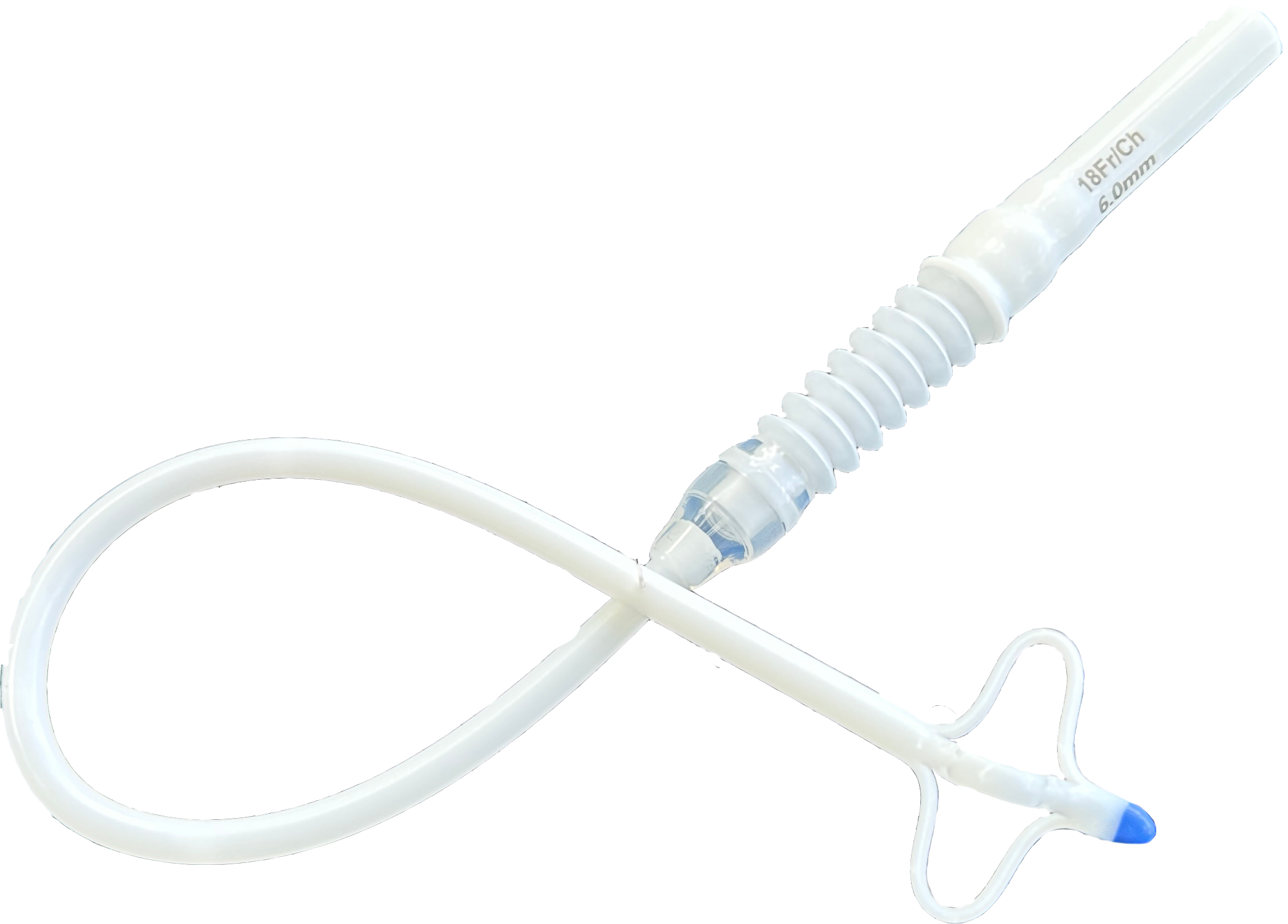
Lotus®
Catheter Balloon-Free |

Foley Catheter
Balloon-Based
|
|---|---|---|
| Insertion & Removal | ~4 sec insertion · ~3 sec removal Single step — push to deploy, pull rod to retract. No syringe, no inflation, no separate supplies. 1 | Multi-step procedure Requires syringe for balloon inflation on insertion and deflation on removal. Incomplete deflation risks urethral trauma during extraction. 4 |
| Overall Complication Rate | 0% UTI · 0% hematuria · ~2% spasms Prospective clinical trial, N=50. 1 | 57% reported ≥1 complication Within 30 days — noninfectious complications were 5× more common than infectious. N=2,076. 2 |
| Urinary Tract Infection | 0% UTI (0 of 50) Zero urinary tract infections detected across entire prospective cohort. 1 Zero infections | 5.7% diagnosed UTI 5.7% told they had UTI; 10.5% reported any infectious complication (fever, chills, burning, urgency). 2 Balloon-linked risk |
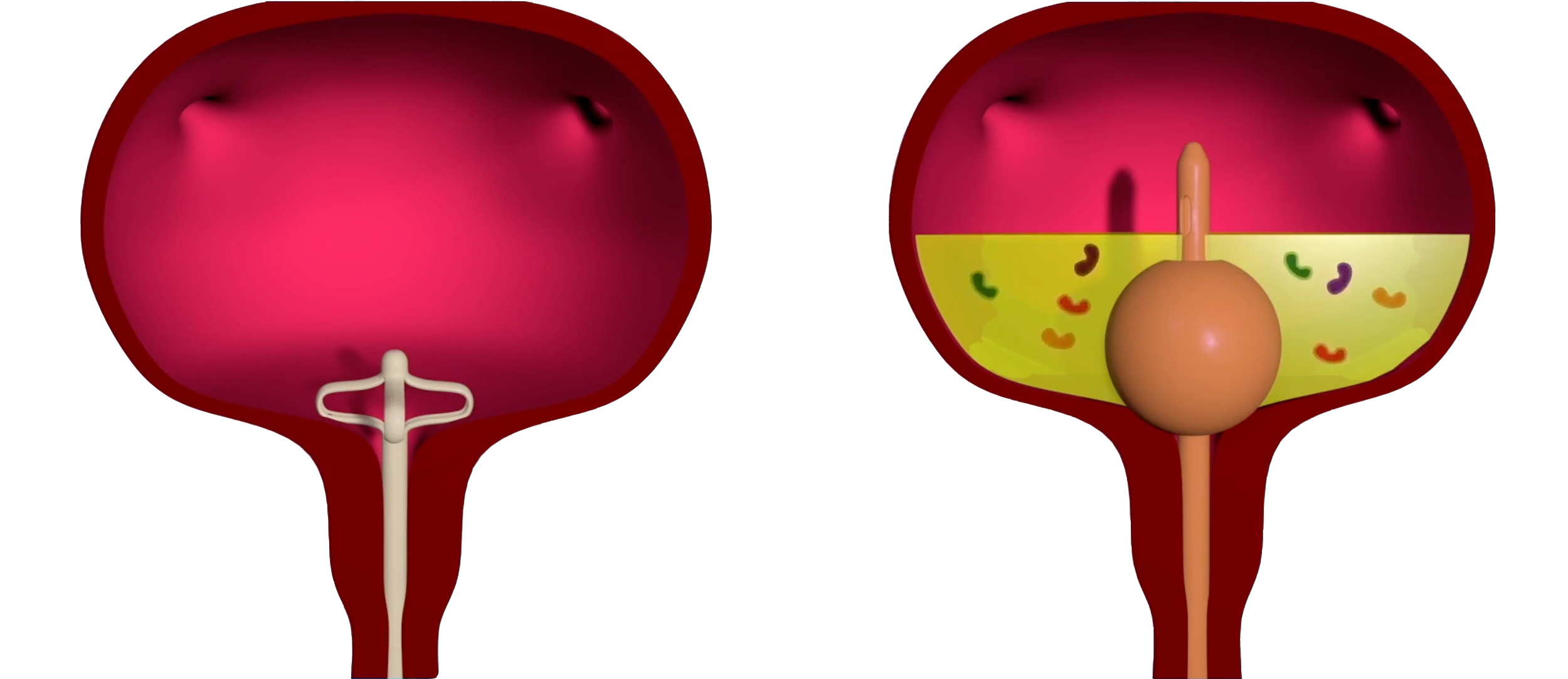
| Residual Urine | 4.8 mL mean PVR Bladder scanner confirmed. PVR >0 in only 8 of 50 patients. Near-complete drainage — lumen rests at bladder neck. 1 Near-complete drainage | 77 mL mean PVR Vs. 0 mL for straight catheters. Balloon elevates lumen above pooled urine, preventing complete emptying and promoting bacterial colonization. 1* Residual urine pool |
| Patient Discomfort | 0/10 insertion · 2/10 removal Wong-Baker scale. Mean insertion discomfort 0 (no pain). Mean removal discomfort 2/10. 1 | 54.5% reported pain Pain or discomfort with catheter in place, patient-reported (n=124 subgroup). 2 |
| Removal Complications | 0% trauma No gross hematuria at insertion. Two accidental dislodgements caused zero urethral injury, no hematuria. 1 Wings compress safely | 30.9% removal complications Composite: pain, discomfort, bleeding, and/or trauma during removal. Separately, 10.0% reported blood in urine while catheterized. 2 Balloon extraction risk |
| Bladder Spasms | 2% (1 of 50) One patient experienced spasms, resolved with short-course anticholinergic medication. May have been procedure-related. 1 | 34.7% with catheter in place 34.7% reported urgency/spasms while catheterized; 24.0% continued post-removal. Balloon pressure on trigone is a recognized contributor. 2 |
| Tissue Contact & Inflammation | 4.5–6.5 mm proximal tip Compressible silicone wings — no sustained rigid pressure on trigone or bladder wall. 7 | Balloon-induced tissue damage Inflated balloon exerts continuous pressure on bladder trigone. Histologic inflammation and damage onset within 6 hours. 3 |
| Accidental Dislodgement | Wings compress through urethra Retention tested at 1 lb (0.45 kg) per ASTM F623. 6 Two clinical cases confirmed zero trauma on dislodgement. 1 | Balloon resists extraction 10 mL balloon requires ~3.8 kg (8.4 lbs) to forcibly remove in female cadavers 5, ~9.3 lbs in males 4 — risks urethral laceration, stricture. |
| Home Use Considerations | Simplified activation Push-to-deploy / pull-to-retract eliminates syringe handling. No risk of intraurethral balloon inflation during self-insertion. 8 | Requires syringe for balloon Self-inflation/deflation adds risk of intraurethral balloon inflation, incomplete deflation, and syringe malfunction. |
| Device Versatility | 3-in-1 device FDA-cleared for indwelling, intermittent, and suprapubic catheterization in a single device. Eliminates need to stock multiple catheter types. 8 | Single function per device Foley (indwelling), straight (intermittent), and suprapubic catheters are separate products with different designs, packaging, and SKUs. |
Near-Complete Emptying. By Design.
The Lotus Catheter's lumen sits low at the trigone — the bladder's natural drainage point — enabling near-complete emptying without a balloon obstructing flow.
A globally recognized urologist and medical device inventor with over five decades in clinical practice. In the early 1980s, Dr. Hakki patented an inflatable penile prosthesis acquired by American Medical Systems (later Pfizer) — a device that has since generated an estimated $1.9 billion in cumulative revenue and restored sexual function for hundreds of thousands of men worldwide.
The Lotus Catheter is his next breakthrough: a balloon-free design built on tens of thousands of patient interactions and a career-long conviction that the Foley's retention mechanism was an unnecessary source of harm.
Reimbursed under existing codes
Insertion is billed under CPT 51702, the same code used for Foley catheterization — no new billing pathways, no prior authorization burden, no workflow disruption.
CPT 51702Designed to be cost-neutral to cost-positive
Priced to compete with Foley catheters while delivering category-defining clinical outcomes. When factoring in CAUTI prevention, reduced CMS penalties, and elimination of anticholinergic prescriptions, Lotus is designed to lower total cost of care.
Manufactured for scale
Produced in ISO 13485-certified facilities with capacity to support national and international distribution. Supply redundancy and quality systems are designed to meet the procurement requirements of large health systems and GPO contracts.
For Clinicians: See the data.
Evaluate Lotus with your team. We provide clinical data packages, product samples for hands-on evaluation, and direct access to our medical team.
- Full clinical study results
- Head-to-head comparison data vs. Foley
- Product evaluation samples
- CAUTI reduction economic modeling
For Partners: Own the future.
Join a global network bringing balloon-free catheter technology to market — through territory-based distribution partnerships or GPO formulary integration.
- Exclusive territory agreements available
- GPO formulary evaluation & value analysis support
- Marketing, clinical training & regulatory dossier assistance
- CMS penalty reduction and CAUTI program alignment
Let's end the balloon era — together.
Whether you're a hospital system evaluating alternatives to the Foley, a GPO exploring formulary consolidation, or a distributor seeking a differentiated portfolio product — we're ready to move quickly.
team@lotus-catheter.com


